Continuous Manufacturing
From Powder to the Coated Tablet
From Batch to
Continuous Manufacturing
Continuous Manufacturing in the Pharmaceutical Industry
Change the production of oral solid dosage forms in the pharmaceutical industry with L.B. Bohle. We support you with our machines and processes on your way to introducing Continuous Manufacturing within your company. Whether you are converting a batch process or introducing a new product, we offer the best solutions and applications.

- Wet Granulation
- Dry Granulation
- Direct Compression
we offer “bin-to-bin” solutions and individual components (modules) for continuous process.
Our stand-alone machines for continuous production cover the following process steps
- Twin-screw Granulation
- Continuous Drying
- Dry Granulation
- Tablet Coating
All of our systems can be complemented by modules from selected and qualified third-party suppliers.
Continuous Manufacturing – Definition
Continuous Manufacturing vs. Batch Manufacturing
Continuous manufacturing is a process of producing pharmaceutical products from start to end in a continuous, non-stop production flow.
In contrast to batch manufacturing where materials must be transported from one single process to the next process, tested and re-fed, in continuous manufacturing all testing, feeding, and processing is done in-line. Sophisticated process using analytical technologies ensure in-process quality.
Continuous manufacturing in the pharmaceutical oral solid dosage industry offers substantial benefits, e.g., increased quality, cost savings, flexibility, operator safety and advantages in research and development. A 24/7 processing time is possible. All these advantages are so substantial, that the US Food and Drug Administration (FDA) promotes the development and introduction of continuous process concepts actively in pharmaceutical manufacturing.
Continuous Manufacturing – Process
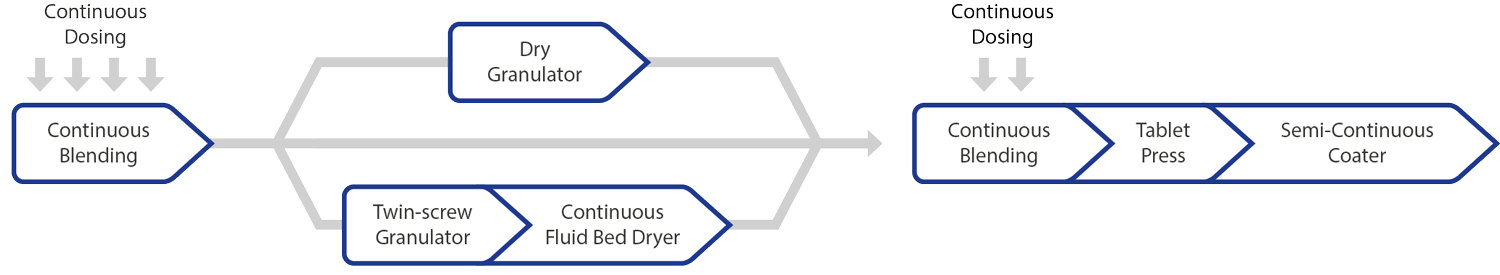
Continuous Manufacturing with QbCon®– Advantages of Continuous Manufacturing
- Best quality - maximum control of the process by using PAT (Process Analytical Technology)
- Integrated processes – multiple plants in one integrated plant
- Shorter production times
- Flexible production – flexible batch sizes and real-time release possible
- Faster research and development cycles – no scale-up problems
- Reduced GMP space requirements
- Higher operator and environmental safety
- Potential savings in
- Personnel / working costs
- Energy consumption
- Resource consumption
- API consumption
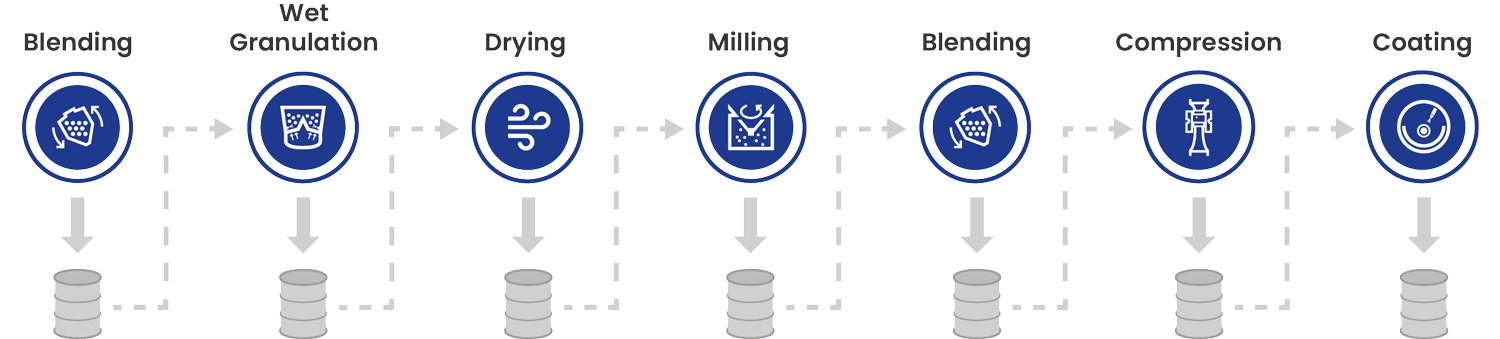
Batch Manufacturing – Definition
All materials are charged before the process starts and discharged at the end of each single process. A batch is a specific quantity intended to have a uniform character and quality within specified limits and produced according to a single manufacturing order in the same manufacturing cycle.
Furthermore, a batch refers to the quantity of material and does not specify the method of production. The product is collected after each unit operation and must be transported to the next process. The finished product is tested in offline laboratories after processing is completed so that processing times can take days or weeks.
Batch Manufacturing – Process
Your Contact for
Continuous Manufacturing
Burkhard Schmidt
Sales Director
inquiry@lbbohle.de
+49 2524 – 93 23 0

